This chapter was copied with permission from Nick Strobel’s Astronomy Notes. Go to his site at www.astronomynotes.com for the updated and corrected version.
Electromagnetic Radiation (Light)
General properties of light, definition of frequency, spectrum, temperature. Light production: Continuous (thermal) spectra, emission lines, absorption lines, and the Bohr model for the atom. Doppler effect and why spectral lines must be used to measure the Doppler shifts.
7.1 Introduction
At least 95% of the celestial information we receive is in the form of light. Because of this fact, astronomers have devised many techniques to decode as much as possible the messages that are encoded in the often extremely faint rays of light. These messages include information about the object’s temperature, motion, chemical composition, gas density, surface gravity, shape, structure, and more! Roughly 85% of the information in light is uncovered by using spectroscopy—spreading the light out into its different constituent colors or wavelengths and analyzing the spectrum. The first part of this chapter covers the characteristics of all forms of light and the following sections cover spectroscopic analysis. The vocabulary terms are in boldface.
7.2 Electric and Magnetic Fields
In order to understand light, you first need to have an understanding of electric fields and magnetic fields. Electrical charges and magnets alter the region of space around them so that they can exert forces on distant objects. This altered space is called a force field (or just a field). Rather than describing the action of forces by having a distant object somehow reach out across space and push or pull on a body, the body simply responds to its local environment. An electric charge or a magnet responds to the field immediately surrounding it. That field is produced by a distant object. In the same way, a massive object can produce a gravity field that distant objects will respond to.
Scientists have known since the early part of the 19th century that electrical fields and magnetic fields are intimately related to each other and applications of this connection are found all around you. Moving electric charge (electric current) creates a magnetic field. Coils of wire can be used to make the large electromagnets used in car junk yards or the tiny electromagnetics in your telephone receiver. Electric motors used to start your car or spin a computer’s harddisk around are other applications of this phenomenon. In fact, ordinary magnets are produced from tiny currents at the atomic level.
A changing magnetic field creates electrical current — an electric field. This concept is used by power generators — large coils of wire are made to turn in a magnetic field (by falling water, wind, or by steam from the heating of water by burning coal or oil or the heat from nuclear reactions). The coils of wire experience a changing magnetic field and electricity is produced. Computer disks and audio and video tapes encode information in magnetic patterns of alternating magnetic directions and magnetic strengths. When the magnetic disk or tape material passes by small coils of wire, electrical currents (electric fields) are produced.
James Clerk Maxwell (lived 1831–1879) put these ideas together and proposed that if a changing magnetic field can make an electric field, then a changing electric field (from an oscillating electric charge, for example) should make a magnetic field. A consequence of this is that changing electric and magnetic fields should trigger each other and these changing fields should move at a speed equal to the speed of light. To conclude this line of reasoning, Maxwell said that light is an electromagnetic wave. Later experiments confirmed Maxwell’s theory.

Electric and magnetic fields oscillate together but perpendicular to each other and the electromagnetic wave moves in a direction perpendicular to both of the fields.
7.3 Properties of Light
Light, electricity, and magnetism are manifestations of the same thing called electromagnetic radiation. The energy you see coming out of the computer screen you are using to read this page is made of fluctuating electric and magnetic energy fields. The electric and magnetic fields oscillate at right angles to each other and the combined wave moves in a direction perpendicular to both of the electric and magnetic field oscillations. This energy also comes in many forms that are not detectable with our eyes such as infrared (IR), radio, X-rays, ultraviolet (UV), and gamma rays.
We feel infrared light as heat and our radios pick up the messages encoded in radio waves emitted by radio stations. Ultraviolet light has high enough energy to damage our skin cells, so our bodies will produce a darker pigment in our skin to prevent exposure of the deeper skin cells to the UV (we tan as a defense mechanism). The special bulbs called “black lights” produce a lot of UV and were used by hospitals to kill bacteria, amoebas, and other micro-organisms. X-rays are produced by very hot things in space. X-rays have more energy than UV, so they can pass through skin, muscles, and organs. They are blocked by bones, so when the doctor takes your X-ray, the picture that results is the shadow image of the X-rays that passed through your body. Because X-rays have such high energy, they can damage or kill cells. A few brief exposures to low-intensity X-rays is okay. The X-ray technician would be exposed to thousands of X-ray exposures if s/he did not use some sort of shielding. Gamma rays are the most energetic form of electromagnetic radiation and are produced in nuclear reactions.
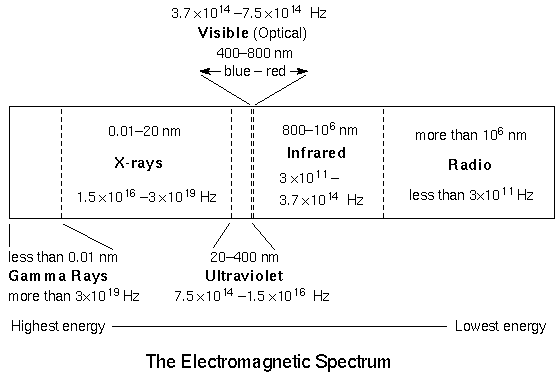
The form of electromagnetic radiation your eyes can detect is called “visible” or “optical.” Astronomers have only recently (within the past few decades) been able to use the other forms of electromagnetic radiation or light. Every time technology has been developed to detect another form of light, a revolution in our understanding of the universe has occurred. The figure above shows all of the forms of electromagnetic radiation in order of INcreasing wavelength (given in nanometers [nm]) and DEcreasing energy. Notice how tiny the visible band is!
There are some general properties shared by all forms of electromagnetic radiation:
- It can travel through empty space. Other types of waves need some sort of medium to move through: water waves need liquid water and sound waves need some gas, liquid, or solid material to be heard.
- The speed of light is constant in space. All forms of light have the same speed of 299,800 kilometers/second in space (often abbreviated as c). From highest energy to lowest energy the forms of light are Gamma rays, X-rays, Ultraviolet, Visible, Infrared, Radio. (Microwaves are high-energy radio waves.)
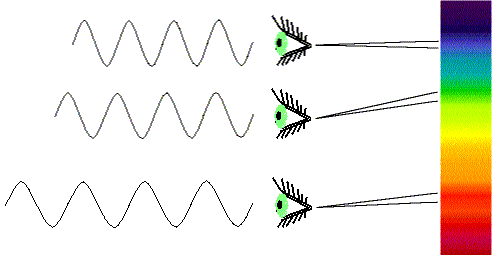
- A wavelength of light is defined similarly to that of water waves—distance between crests or between troughs. Visible light (what your eye detects) has wavelengths 4000-8000 Ångstroms. 1 Ångstrom = 10-10 meter. Visible light is sometimes also measured in nanometers (“nm” in the figure above): 1 nanometer = 10-9 meter = 10 Ångstroms, so in nanometers, the visible band is from 400 to 800 nanometers. Radio wavelengths are often measured in centimeters: 1 centimeter = 10-2 meter = 0.01 meter. The abbreviation used for wavelength is the Greek letter lambda: λ.
White light is made of different colors (wavelengths). When white light is passed through a prism or diffraction grating, it is spread out into all of its different colors. You see this happen every time you see a rainbow. Not all wavelengths of light from space make it to the surface. Only long-wave UV, Visible, parts of the IR and radio bands make it to surface. More IR reaches elevations above 9,000 feet (2765 meters) elevation. That is one reason why modern observatories are built on top of very high mountains. Fortunately, as far as life is concerned, our atmosphere shields us from the gamma rays, X-rays, and most of the UV. It also blocks most of the IR and parts of the radio. Astronomers were not able to detect these forms of energy from celestial objects until the space age, when they could put satellite observatories in orbit.
Besides using wavelength to describe the form of light, you can also use the frequency–the number of crests of the wave that pass by a point every second. Frequency is measured in units of hertz (Hz): 1 hertz = 1 wave crest/second. For light there is a simple relation between the speed of light (c), wavelength (λ), and frequency (f):
f = c/λ.
Since the wavelength λ is in the bottom of the fraction, the frequency is inversely proportional to the wavelength. This means that light with a smaller wavelength has a higher (larger) frequency. Light with a longer wavelength has a lower (smaller) frequency.
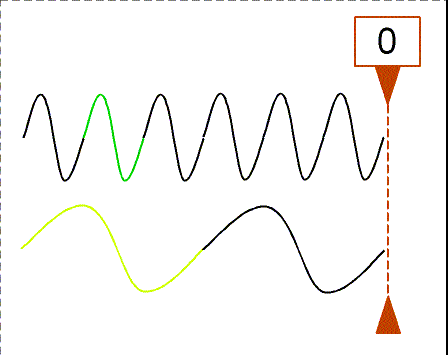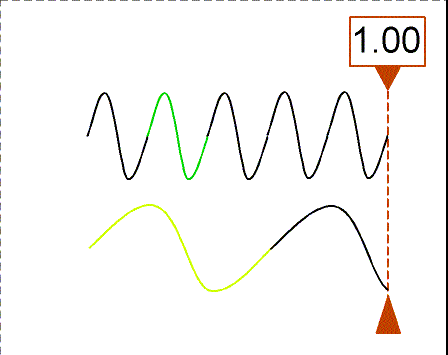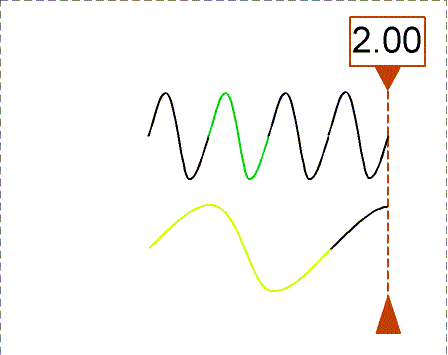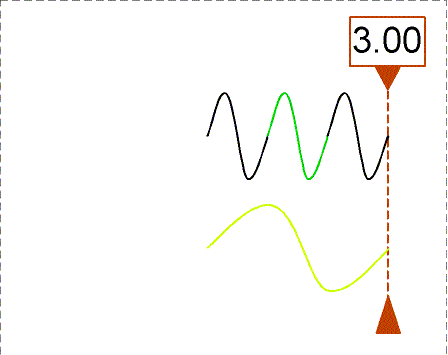
The animation shows waves with different wavelengths moving to the right with the same speed. The bottom wave has a wavelength = 3×(wavelength of the top wave). The counter shows how many wavelengths of the top wave have passed the dashed line. In one second, the top wave moves three wavelengths to the right so its frequency is 3 Hz. The bottom wave moves one of its wavelengths in one second so its frequency is 1 Hz (= 1/3×top wave frequency).
Some colors and their approximate wavelength, frequency and energy ranges are given in the table below. The unit of energy is the Joule (J). A Joule is how much energy you expend when you lift an object with 1 kilogram of mass (for example, a liter of water) about 10 centimeters above the ground. If you then let it go, the object hits the ground with that much energy. Sometimes light energy is also measured in “ergs,” where 1 erg = 10-7 Joule.
| color | f (*1014 Hz) | Energy (*10-19 J) | |
|---|---|---|---|
| violet | 4000 |
7.5 |
5.0 |
| indigo | 4600 |
6.5 |
4.3 |
| blue | 4750 |
6.3 |
4.2 |
| green | 4900 |
6.1 |
4.1 |
| yellow | 5650 |
5.3 |
3.5 |
| orange | 5750 |
5.2 |
3.45 |
| red | 6000 |
5.0 |
3.3 |
Note the trends: bluer light has shorter λ, higher f, and more energy. Redder light has longer λ, lower f, and less energy.
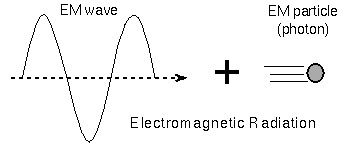
At the beginning of the 20th century Max Planck (lived 1858–1947) suggested that atoms can absorb and emit energy in only discrete chunks (called quanta). This quantum behavior of atoms could explain the drop-off of a continuous spectrum’s shape at the short wavelength end. A few years after Planck’s discovery Albert Einstein (lived 1879–1955) discovered that the quantum of energy was not due to the atoms but, rather, a property of the energy itself. You can consider light as packets of energy called photons. A photon is a particle of electromagnetic radiation. Bizarre though it may be, light is both a particle and a wave. Whether light behaves like a wave or like a particle depends on how the light is observed (it depends on the experimental setup)!
Einstein found a very simple relationship between the energy of a light wave (photon) and its frequency:
Energy of light = h x f
Energy of light = (h x c) / λ
where h is a universal constant of nature called “Planck’s constant” = 6.63 × 10-34 J·sec.
Light can also behave as a particle and a wave at the same time. An example of light acting as both a particle and a wave is the digital camera — the lens refracts (bends and focuses) waves of light that hit a charge-coupled device (CCD). The photons kick electrons out of the silicon in the CCD. The electrons are detected by electronics that interpret the number of electrons released and their position of release from the silicon to create an image. Another example is when you observe the build-up of the alternating light and dark pattern from diffraction (a wave phenomenon) from light passing through a narrow slit. You see one bright spot (a photon), then another bright spot (another photon), then another… until the diffraction pattern is created from all of the accumulated photons. This happens so quickly that it is undetectable to the human eye.
To decode the information stored in light, you pass the light through a prism or diffraction grating to create a spectrum—any display of the intensity of light (EM radiation) at different wavelengths or frequencies (a picture or a graph of intensity vs. either wavelength or frequency). If white light is examined, then the spectrum will be a rainbow.
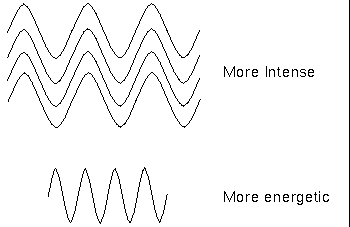
The term intensity has a particular meaning here: it is the number of waves or photons of light reaching your detector; a brighter object is more intense but not necessarily more energetic. Remember that a photon’s energy depends on the wavelength (or frequency) only, not the intensity. The photons in a dim beam of X-ray light are much more energetic than the photons in an intense beam of infrared light.
The type of light produced by an object will depend on its temperature, so let’s digress slightly to investigate what “temperature” is. Temperature is a measure of the random motion (or energy) of a group of particles. Higher temperature (T) means more random motion (or energy). A natural scale would have zero motion at zero degrees (absolute zero). This scale is the Kelvin scale. It scales exactly like the Celsius system, but it is offset by 273 degrees. Here is a comparison of the Kelvin, Celsius, and Fahrenheit temperature scales:
| K | C | F | |
|---|---|---|---|
| 0 | -273 | -459 | absolute zero |
| 100 | -173 | -279.4 | |
| 273 | 0 | 32 | water freezes |
| 310 | 37 | 98.6 | human temperature |
| 373 | 100 | 212 | water boils (STP) |
| 755 | 482 | 900 | oven on “clean” setting |
| 5840 | 5567 | 10053 | Sun’s temperature |
Vocabulary
- electromagnetic radiation
- frequency
- hertz
- intensity
- Kelvin
- photon
- spectroscopy
- spectrum
- temperature
- wavelength
Formulae
- Frequency and wavelength relation: f = c/λ.
- Energy of a photon: E = h×f, where h is a constant of nature.
- Energy of a photon: E = (h×c)/λ.
Review Questions 1
- Why is light so very important to astronomy? What kinds of information can you get from it?
- Why is light called electromagnetic radiation? Is radio a form of light?
- Put the following forms of light in order of increasing frequency (lowest frequency first): ultraviolet, infrared, gamma rays, visible, radio, X-rays. Put them in order of increasing wavelength (shortest wavelength first). Put them in order of increasing energy (lowest energy first).
- Do all forms of light travel at the same speed in a vacuum (empty space)? Why is it important that light can travel through empty space?
- What forms of light can be observed from the ground (including high mountains)? What forms can be observed at high altitudes in our atmosphere? What forms must be observed in space?
- Is electromagnetic radiation a wave or a particle? What determines if you will see light as a wave or a photon?
- Which of these are a spectrum: plot of intensity vs. wavelength, plot of intensity vs. brightness, plot of frequency vs. wavelength, rainbow, plot of acceleration vs. time, plot of energy vs. frequency.
- What is the difference between intensity and energy? If a particular CCD chip requires light energies of 4.2 × 10-19 J to release electrons from the silicon, which will produce more electrons (and hence, a brighter computer image): an intense beam of yellow light or a dim beam of UV light? Explain your answer!
- Why is the Kelvin scale preferred over the Celsius or the Fahrenheit scales?
- Where is absolute zero on the three temperature scales? Where is the Sun’s temperature on the three temperature scales?
7.4 Production of Light
When light is passed through a prism or a diffraction grating to produce a spectrum, the type of spectrum you will see depends on what kind of object is producing the light: is it a thick or thin gas, is it hot or cool, is it a gas or a solid? There are two basic types of spectra: continuous spectrum (energy at all wavelengths) and discrete spectrum (energy at only certain wavelengths). Astronomers usually refer to the two types of discrete spectra: emission lines (bright lines) and absorption lines (dark lines in an otherwise continuous spectrum) as different types of spectra.
7.4.1 Continuous Spectrum
A rainbow is an example of a continuous spectrum. Most continuous spectra are from hot, dense objects like stars, planets, or moons. The continuous spectrum from these kinds of objects is also called a thermal spectrum, because hot, dense objects will emit electromagnetic radiation at all wavelengths or colors. Any solid, liquid and dense (thick) gas at a temperature above absolute zero will produce a thermal spectrum. A thermal spectrum is the simplest type of spectrum because its shape depends on only the temperature. A discrete spectrum is more complex because it depends on temperature and other things like the chemical composition of the object, the gas density, surface gravity, speed, etc. Exotic objects like neutron stars and black holes can produce another type of continuous spectrum called “synchrotron spectrum” from charged particles swirling around magnetic fields, but I will discuss them in another chapter later on. For now, let’s look at a thermal spectrum.
Sometimes astronomers use the term “blackbody” spectrum for a thermal spectrum. A “blackbody” is an object that absorbs all the light falling on it, reflecting none of it, hence, it appears black. When the “blackbody” object is heated, it emits light very efficiently without any gaps or breaks in the brightness. Though no object is a perfect “blackbody,” most stars, planets, moons and asteroids are near enough to being “blackbodies,” that they will produce spectra very similar to a perfect thermal spectrum.

Some thermal spectra for objects of different temperatures are illustrated in the figure below.
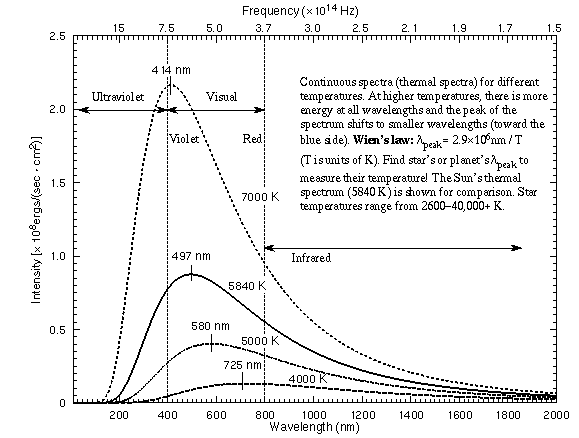
Some key features of a thermal (continuous) spectrum are as follows:
- There is light from a dense object at all possible λ IF the object is above 0 K (absolute zero). Since everything in the universe is above 0 K, all dense objects (solids, liquids, thick gases) will produce a thermal spectrum.
- The shape of a continuous spectrum depends on only the temperature of the object NOT its chemical composition. This allows you to determine the temperature of an object from a great distance away.
- As the temperature of an object increases, more light is produced at all wavelengths than when it was cooler. You can see this effect with a light bulb wired to a dimmer switch. As you raise the current going to the bulb, the bulb’s filament gets hotter and brighter.
- As the temperature of an object increases, the peak of thermal spectrum curve shifts to smaller wavelengths (higher frequencies) — cool things appear red or orange, hotter things appear yellow or white, and very hot things blue or purple. This is opposite to what artists use for “cool” colors (blues) or “hot” colors (reds)! You can also see this effect with the light bulb wired to a dimmer switch. The dim bulb will have an orange color and as you make it brighter, the bulb will turn yellow and even white.
 Wilhelm Wien (lived 1864–1928) discovered that the peak of the thermal spectrum curve, λpeak in nanometers, is related to the temperature by λpeak = 2.9 × 106 / temperature (in K). This simple relation is now known as Wien’s Law. Using this you will find that cool objects like cars, plants, and people radiate most of their energy in the infrared. Very cold objects radiate mostly in the radio band.
Wilhelm Wien (lived 1864–1928) discovered that the peak of the thermal spectrum curve, λpeak in nanometers, is related to the temperature by λpeak = 2.9 × 106 / temperature (in K). This simple relation is now known as Wien’s Law. Using this you will find that cool objects like cars, plants, and people radiate most of their energy in the infrared. Very cold objects radiate mostly in the radio band.- A small change in the temperature produces a HUGE change in the amount of energy emitted by every unit area of the object. If you add up all of the energy emitted every second by an area of one square meter on the object’s surface, you find it equals σ × temperature4, where σ is another universal constant of nature [= 5.67×10-8 J/(m2 K4 s)]. This relation is called the Stefan-Boltzmann law. Because the temperature is raised to the fourth power, a small rise in the temperature of an object will produce a HUGE increase in the amount of energy it emits.
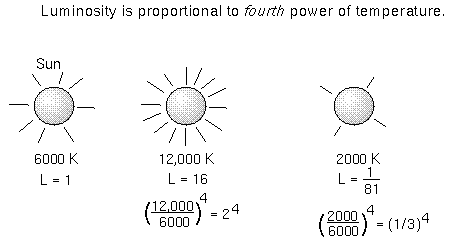
When you add up all of the energy of all of the square meters on the object’s surface, you get the luminosity—the total amount of energy emitted every second by the object. The luminosity = (total surface area) × (σ × temperature4). If our Sun were just twice as hot as it is now, it would produce 24 = 16 times more energy than it does now!
The UNL Astronomy Education program’s Blackbody Curves module lets you explore the relationship between temperature and the thermal spectrum by manipulating various parameters with a graphical interface (link will appear in a new window). It also introduces the concept of filters.
7.4.2 Discrete Spectrum
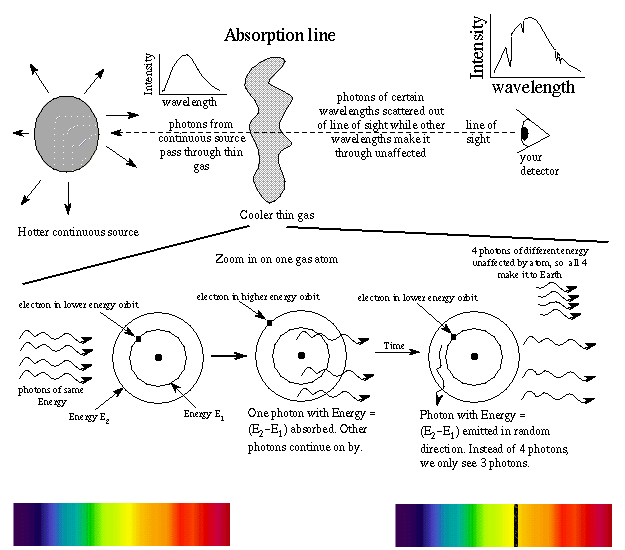
Close examination of the spectra from the Sun and other stars reveals that the rainbow of colors has many dark lines in it, called absorption lines. They are produced when cooler thin gas absorbs certain colors of light produced by hotter dense objects, so for stars, the cooler, low-density upper layers absorb certain colors produced by the deeper, denser layers. The cool thin gas is thin enough that most of the light can pass right through it without hitting any atoms or molecules (e.g., as thin as the upper layers of the Earth’s atmosphere many tens of kilometers above the Earth’s surface). You can also see them in the reflected light spectrum from planets. Some of the colors in the sunlight reflecting off the planets are absorbed by the molecules on the planet’s surface or in its atmosphere.
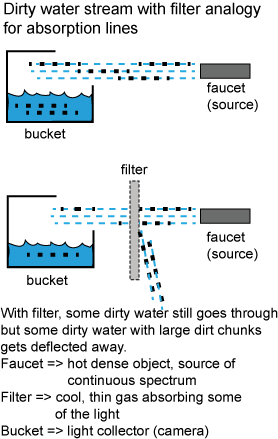
The process in making absorption lines is analogous to putting a filter in a narrow stream of dirty water that is collecting in a bucket. Without the filter, all of the dirty water and dirt chunks collect in the bucket. With the filter, some of the dirty water and the dirt chunks get deflected away from the bucket, so not as much of the dirty water and dirt chunks make it into the bucket. In the analogy, the source of the continuous spectrum light is like the faucet spewing out the dirty water, the cool thin gas is like the filter, and the bucket is like the camera or telescope. What is not in the analogy is the spectrometer that takes the collected light and spreads it out into its constituent colors.
The spectra of hot, thin (low density) gas clouds are a series of bright lines called emission lines. In both of these types of spectra you see spectral features at certain, discrete wavelengths (or colors) and no where else.
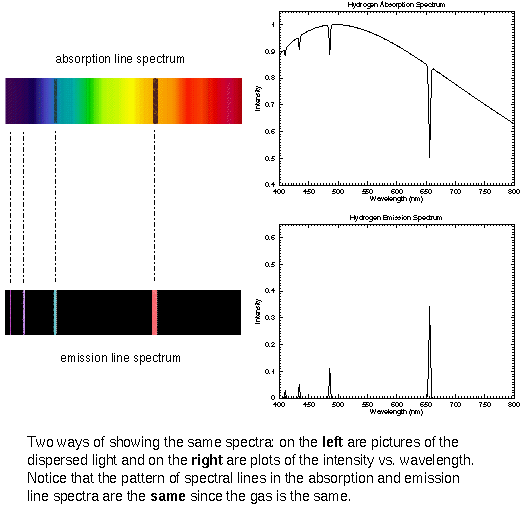
The type of spectrum you see depends on the temperature of the thin gas. If the thin gas is cooler than the thermal source in the background, you see absorption lines. Since the spectra of stars show absorption lines, it tells you that the density and temperature of the upper layers of a star is lower than the deeper layers. In a few cases you can see emission lines on top of the thermal spectrum. This is produced by thin gas that is hotter than the thermal source in the background. Unlike the case for absorption lines, though, the production of emission lines does NOT require a thermal source be in the background. The spectrum of a hydrogen-emission nebula (“nebula” = gas or dust cloud) is just a series of emission lines without any thermal spectrum because there are no stars visible behind the hot nebula. Some objects produce spectra that is a combination of a thermal spectrum, emission lines, and absorption lines simultaneously!
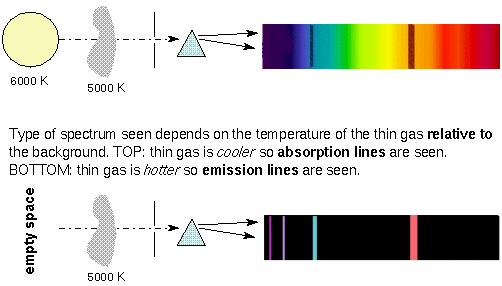
What is very useful about discrete spectra is that the pattern of lines you see depends on the chemical composition of the thin gas. Each element or molecule produces a distinct pattern of lines—each element or molecule has a “fingerprint” you can use to identify it. This allows you to remotely determine what stars, planets, nebulae, etc. are made of!
The composition canNOT be found from just one line because one element may have one spectral line at the same wavelength as another element’s spectral line. However, an element’s pattern of lines is unique. Using a single line to identify a gas would be like identifying the name of someone using just one letter of their name—many people will have that same letter in their name, but the pattern of letters (which letters and how they are arranged) is unique to that one person. Of course, stars, planets, nebulae, etc. are made of more than one type of material, so you see the discrete spectra of many elements and molecules superimposed on each other—all of the spectral lines add together. An experienced astronomer can disentangle all the different patterns and sort out the elements and molecules (but it does take time!).
A couple of nice interactives to try matching spectral lines to chemical composition are
- The Science of Light’s Stellar Spectra lab at Learner.org (selecting the link will bring up the site in a new window either in front of or behind this window).
- Decoding Cosmic Spectra from Nova’s Origins series that was broadcast on PBS. (Selecting the link will bring it up in a new window).
Sections Review
Vocabulary
- absorption line spectrum
- continuous spectrum
- discrete spectrum
- emission line spectrum
- luminosity
- Stefan-Boltzmann law
- thermal spectrum
- Wien’s law
Formulae
- Wien’s Law: λpeak = 2.9 × 106/temperature. The units of the peak wavelength are nanometers and the temperature is in Kelvin.
- Stefan-Boltzmann Law: Energy emitted by a square meter on an object’s surface = σ × temperature4, where σ is a constant of nature.
Review Questions 2
- What are the three basic kinds of spectrum? Can an object produce more than one type at the same time?
- What produces a thermal spectrum? Does it depend on chemical composition?
- How can temperature be determined from a continuous spectrum? How would the color of a hot object compare to the color of a cooler object? At what wavelength do you at 98.6 degrees Fahrenheit radiate the most? (Hint: use the temperature scales table.)
- How will the thermal spectrum produced by a chunk of lead compare to the thermal spectrum produced by a chunk of iron of the same size and temperature?
- What produces an emission line spectrum? Do you need a thermal source in the background?
- Can you see emission lines if a thermal source is in the background? What does their visibility depend on? (Think about the temperature of the gas producing the emission lines and the temperature of the the background thermal source.)
- What produces an absorption line spectrum? Do you need a thermal source? Would you see absorption lines if the gas in front of a thermal source was hotter than the thermal source? Explain why.
- Why must you use a pattern of lines to find the composition? Why is one line not sufficient?
- What kind of spectrum and what pattern of lines would you see if you heated up a tube filled with hydrogen, helium and neon gas?
7.5 Bohr atom
Scientists have had the technology to observe discrete spectra since the beginning of the 19th century. They had to wait over a hundred years, though, for an explanation of how the discrete spectra were produced. They knew that it was produced by atoms and that atoms had negative and positive charges in them. Some models of the atom were similar to our current one: the positive charges are concentrated in a central nucleus with the negative charges swarming around it, but the atoms should be unstable. As the negative charges (called electrons) move around the nucleus, they should radiate light and spiral into the nucleus in about 10-16 second. This is obviously contradicted by common experience!
Niels Bohr (lived 1885–1962) provided the explanation in the early 20th century. He said that the electron can be only found in energy orbits of a certain size and as long as the electron is in one of those special orbits, it would radiate no energy. If the electron changed orbits, it would radiate or absorb energy. This model sounds outlandish, but numerous experiments have shown it to be true.
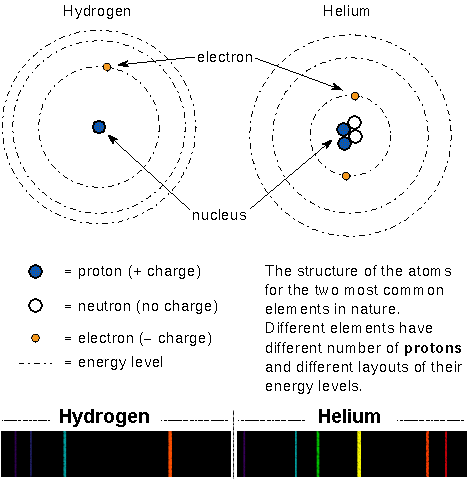
In Bohr’s model of the atom, the massive but small positively-charged protons and massive but small neutral neutrons are found in the tiny nucleus. The small, light negatively-charged electrons move around the nucleus in certain specific orbits (energies). In a neutral atom the number of electrons = the number of protons. The arrangement of an atom’s energy orbits depends on the number of protons and neutrons in the nucleus and the number of electrons orbiting the nucleus. Because every type of atom has a unique arrangement of the energy orbits, they produce a unique pattern of absorption or emission lines.
Select here for an enlargement of the spectra.
All atoms with the same number of protons in the nucleus are grouped together into something called an element. Because the atoms of an element have the same number of protons, they also have the same number of electrons and, therefore, the same chemical properties. For example, all atoms with one proton in the nucleus have the same chemical properties and are called Hydrogen. All atoms with two protons in the nucleus will not chemically react with any other atoms and are known as Helium. The atoms called Carbon form the basis of life and have six protons in the nucleus. In the figure below, atom (a) is Hydrogen, atom (b) is Helium, atoms (c), (d), and (e) are Lithium.

Elements are sub-divided into sub-groups called isotopes based on the number of protons AND neutrons in the nucleus. All atoms of an element with the same number of neutrons in the nucleus are of the same type of isotope. An element’s isotopes will have very nearly the same chemical properties but they can behave very differently in nuclear reactions. For example, all of the isotopes of the element Hydrogen have one electron orbiting the nucleus and behave the same way in chemistry reactions. The ordinary Hydrogen isotope has 0 neutrons + 1 proton while another Hydrogen isotope called Deuterium has 1 neutron + 1 proton and another Hydrogen isotope called Tritium has 2 neutrons + 1 proton in the nucleus. Tritium is radioactive — its nucleus spontaneously changes into another type of nucleus. In the figure above, atoms (c), (d), and (e) are different isotopes of the same element called Lithium.
Most atoms in nature are neutral, the negative charges exactly cancel the positive charges. But sometimes an atom has a hard collision with another atom or absorbs an energetic photon so that one or more electrons are knocked out of the atom. In some rare cases, an atom may temporarily hold onto an extra electron. In either case, the atom has an extra positive or negative charge and is called an ion. For example, the carbon ion C+ has 6 protons and 5 electrons and the iron ion Fe2+ has 26 protons and 24 electrons. Because the number of electrons are different, an ion of an element will behave differently in chemical reactions than its neutral cousins. In the figure above atom (d) is a Li+ ion [compare it with atom (c) or (e)].
In order to explain discrete spectra, Bohr found that atoms obey three basic rules:
- Electrons have only certain energies corresponding to particular distances from nucleus. As long as the electron is in one of those energy orbits, it will not lose or absorb any energy. The energy orbits are analogous to rungs on a ladder: electrons can be only on rungs of the ladder and not in between rungs.
- The orbits closer to the nucleus have lower energy.
- Atoms want to be in the lowest possible energy state called the ground state (all electrons as close to the nucleus as possible).
7.6 How Atoms Produce the Spectra
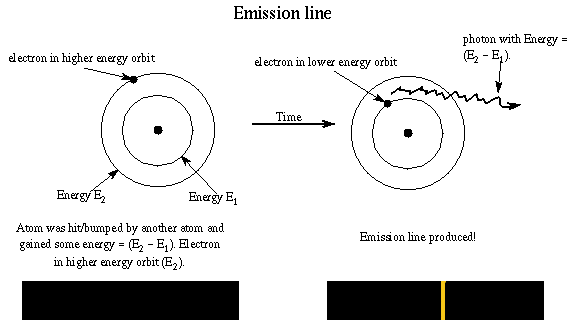
Let’s see how Bohr’s model of the atom explains the three types of spectra. An emission line is produced by an atom in an “excited” energy state — the electron is not in as low an energy orbit as possible. Remember rule #3! In order to go to a lower energy orbit, the electron must lose energy of a certain specific amount. The atom releases the energy is the form of a photon with that particular energy. The energy of photon = the difference in energy of the energy orbits (energy ladder rungs).
Example: An atom with an electron at the E2 orbit and wants to get to the lower E1 energy orbit. It gives off a photon with energy E = h × f = E2 – E1. The electron may reach the ground state in one jump or it may temporarily stop at one or more energy levels on the way, but it canNOT stop somewhere between the energy levels. Different jumps produce photons of different energies. A larger jump to a lower energy level, will produce a photon with greater energy (smaller wavelength).
The atom produces light of certain wavelengths. (Remember that light is both a photon and a wave!) The more atoms undergoing a particular transition, the more intense the emission line will be. The intensity depends on the density and temperature of the gas.
An absorption line is produced when a photon of just the right energy is absorbed by an atom, kicking an electron to a higher energy orbit. The photon had energy = the difference in energy of the energy orbits. Because the energy levels in an element’s atoms are fixed, the size of the outward jumps made by the electrons are the same as the inward jumps. Therefore, the pattern of absorption lines is the same as the pattern of emission lines. Other photons moving through the gas with the wrong energy will pass right on by the atoms in the thin gas. They make up the rest of the continuous spectrum you see.
Example: An atom with electron in the E1 orbit sees a photon with energy Ephoton = E2 – E1. The photon is absorbed and electron moves to E2. The photon is later re-emitted but in a random direction—not necessarily in the same direction as the original photon! An observer will see less photons from the direction of the continuous source at that specific frequency (color) than other frequencies (colors). Photons of other energies pass right on by without being absorbed. The atom can absorb photons of just the right energy to move an electron from one energy level to another level. The more atoms undergoing a particular absorption transition, the darker (or “stronger”) the absorption line. The strength of the absorption line depends on the density and temperature.
A thermal spectrum is produced by atoms that are closely packed together. The energy levels of the atoms are distorted by their neighboring atom’s electrons. This smears out the normally sharp spectral lines (they become fatter).
Example: An orange line is fattened so that one edge is in the yellow wavelengths and the other edge is in the red wavelengths. The amount of smearing, or broadening, depends on the density. Eventually, the density gets high enough to where the smeared lines all merge together to produce the rainbow of colors of a continuous spectrum.
Use the UNL Astronomy Education program’s Hydrogen Energy Levels module to further explore how emission and absorption lines are created (link will appear in a new window). You can also see how the number of atoms in a given state (electrons in a given energy level) changes with the temperature.
7.7 Universality of Physical Laws in Space and Time
The pattern of spectral lines and particular wavelengths produced by an atom depend very sensitively on the masses and charges of the sub-atomic particles and the interactions between them (forces and rules they follow). If different parts of the universe had even slight differences in the rules of quantum mechanics that govern the interactions of the protons, electrons, and neutrons or differences in the strengths of the fundamental forces of nature from that observed on the Earth, we would see noticeable changes in the spacing and strength of the spectral lines. If the subatomic particles had different amount of charge or mass, the pattern of lines would be different than what you see on the Earth. We observe that all hydrogen atoms produce the same pattern of lines everywhere in the universe, whether they are in the Sun, other stars, distant galaxies, or intergalactic space. (Hydrogen is not special in this regard — all of the other types of atoms produce the same pattern that is unique to that type of atom.) The fact that we see the same pattern of lines for a given element (type of atom) everywhere in the universe tells us that the same laws of physics used in the structure of atoms work everywhere in the universe.
Recall from the first chapter that astronomy gives us a sort of time machine: when we look at very distant objects we see them as they were a long time ago because light, traveling at a fast but finite speed, has taken a long time to travel the vast distances between us and the distant objects. The light from those distant objects tells us about the physical laws operating in that part of the universe when the light was emitted long ago. The fact that we see the same pattern of lines for a given element in light emitted by objects at any time in the universe tells us that the laws of physics are the same throughout time — they do not change.
You will also see in the following chapters that the same kind of conclusions can be reached by looking at the gravitational interactions of objects at any position and distance from us. We see the same laws of nature operating at all places and times in the universe. If you reflect a bit on that fact, it is pretty darn amazing!
Sections Review
Vocabulary
- electron
- element
- ground state
- ion
- isotope
- neutron
- proton
Review Questions 3
- Where are electrons, protons, and neutrons located in the atom? Can the electron be found at any position or energy in the atom?
- How does the Bohr atom model explain emission line spectra?
- Which produces a shorter wavelength of light: an electron jumping from the 6th to 2nd energy level, or one jumping from 3rd to 2nd energy level? Explain your answer.
- How does the Bohr atom model explain absorption line spectra?
- Which would produce an absorption line at a longer wavelength: an electron jumping from the 1st to 5th energy level, or one jumping from 1st to 3rd energy level? Explain your answer.
- Which will produce a stronger absorption line: a 10,000 K cloud with 100 particles in front of a hot star or a 10,000 K cloud with 1,000,000 particles in front of a hot star? Why is that?
- If the atom absorbs a photon and later emits it, why do we see any absorption lines at all?
- How does the Bohr atom model explain thermal spectra?
- How will the spectra from atoms of the hydrogen isotopes deuterium and tritium compare to the spectra of ordinary hydrogen atoms?
- Why would you not expect the absorption lines of the calcium ion Ca+ to be the same as the ones of neutral calcium Ca?
- Will the hydrogen ion H+ produce any absorption lines or emission lines? Explain your answer.
- How do we know that the laws of physics (nature) are the same everywhere in the universe and that they are the same throughout time?
7.8 Doppler Effect
The wave nature of light means there will be a shift in the spectral lines of an object if it is moving. This effect is known as the Doppler effect. You have probably heard the Doppler effect in the change of the pitch of the sound coming from something moving toward you or away from you (e.g., a train whistle, a police siren, an ice cream truck’s music, a mosquito buzzing). Sounds from objects moving toward you are at a higher pitch because the sound waves are compressed together, shortening the wavelength of the sound waves. Sounds from objects moving away from you are at a lower pitch because the sound waves are stretched apart, lengthening the wavelength. Light behaves in the same way.

Motion of the light source causes the spectral lines to shift positions. An object’s motion causes a wavelength shift Δλ = λnew – λrest that depends on the speed and direction the object is moving. The amount of the shift depends on the object’s speed: Δλ = λrest × Vradial / c, where c is the speed of light, λrest is the wavelength you would measure if the object was at rest and Vradial is the speed along the line of sight.
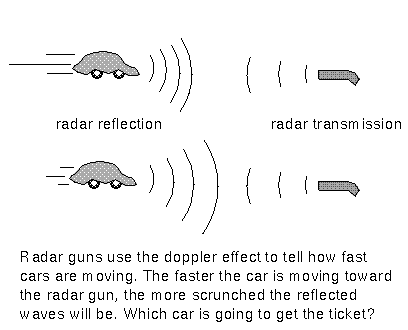
There is a lot of information stored in that little formula! First, it says that the faster the object moves, the greater the Doppler shift Δλ. For example, a particular emission line of hydrogen from nearby galaxies is shifted by a smaller amount than the same line from faraway galaxies. This means that the faraway galaxies are moving faster than the nearby galaxies. The “radar guns” used by police officers operate on this principle too. They send out a radio wave of a set wavelength (or frequency) that reflects off a car back to the “radar gun.” The device determines the car’s speed from the difference in the wavelength (or frequency) of the transmitted beam and reflected beam.
Second, the term Vradial means that only the object’s motion along the line of sight is important. If object moves at an angle with respect to the line of sight, then the Doppler shift (Δλ) tells you only about the part of its motion along the line of sight. You must use other techniques to determine how much of an object’s total velocity is perpendicular to the line of sight.
Finally, which way the spectral lines are shifted tells you if the object is moving toward or away from you. If the object is moving toward you, the waves are compressed, so their wavelength is shorter. The lines are shifted to shorter (bluer) wavelengths — this is called a blueshift. If the object is moving away from you, the waves are stretched out, so their wavelength is longer. The lines are shifted to longer (redder) wavelengths—this is called a redshift.
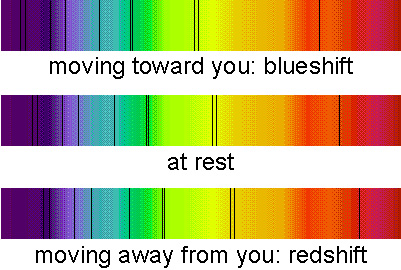
This explanation also works if you are moving and the object is stationary or if both you and the object are moving. The Doppler effect will tell you about the relative motion of the object with respect to you. The spectral lines of nearly all of the galaxies in the universe are shifted to the red end of the spectrum. This means that the galaxies are moving away from the Milky Way galaxy and is evidence for the expansion of the universe.
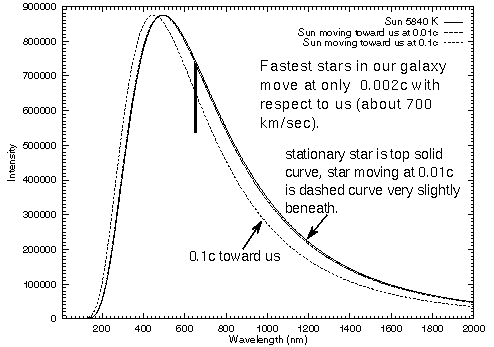
The Doppler effect will not affect the overall color of an object unless it is moving at a significant fraction of the speed of light (VERY fast!). For an object moving toward us, the red colors will be shifted to the orange and the near-infrared will be shifted to the red, etc. All of the colors shift. The overall color of the object depends on the combined intensities of all of the wavelengths (colors). The first figure below shows the continuous spectra for the Sun at three speeds (zero, a fast 0.01c, a VERY fast 0.1c). The Hydrogen-alpha line (at 656.3nm) is shown too. Objects in our galaxy move at speeds much less than 0.01c. The Doppler-shifted continuous spectrum for the Sun moving at 0.01c is almost indistinguishable from the Sun at rest even when you zoom in to just the optical wavelengths (second figure). However, the Doppler shift of the spectral line is easy to spot for the slow speed. By zooming in even further, you can detect spectral line Doppler shifts for speeds as small as 1 km/sec or lower (less than 3.334×10-6 c).
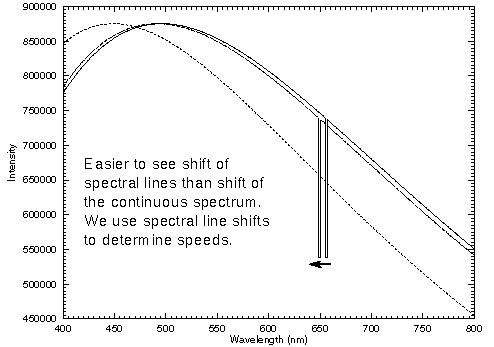
Vocabulary
- blueshift
- Doppler effect
- redshift
Formulae
- Wavelength shift: Δλ = λnew – λrest, where λrest is the wavelength measured if the object is at rest and λnew is the wavelength measured for the moving object.
- Doppler effect: Δλ = λrest × Vradial/c, where Vradial is the object’s speed along the line of sight and c is the speed of light.
- If λnew > λrest, the object is moving away (redshift).
- If λnew < λrest, the object is approaching (blueshift).
Review Questions 4
- What two things can the Doppler effect tell you about an object’s relative motion?
- Which galaxy is moving faster: one with lines red-shifted by 200 km/s or one blue-shifted by 300 km/s? Which one is moving away from us?
- Would a star moving left to right with respect to you have a Doppler shift? Why or why not?
- There are two edge-on binary systems (orbiting in a plane along our line of sight) with the two stars separated by 2 AU. Which will produce the largest shifting of their spectral lines: the system with star masses 10 & 15 solar masses or the system with 1.5 & 2.0 solar masses? Explain why.
- There are two binary systems with the two stars separated by 2 AU. Which will produce the largest shifting of their spectral lines: an edge-on system with star masses 1.5 & 2.0 solar masses or a face-on system (stars orbiting in a plane perpendicular to our line of sight) with star masses 10 & 15 solar masses? Explain why.
- Why should you use spectral lines to measure velocities rather than the change in color of an object?



